Atom From The Element Carbon Zdarma
Atom From The Element Carbon Zdarma. Where more than one isotope exists, the value given is the abundance weighted average. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. In its diamond allotrope, carbon is the hardest known substance in nature. Three naturally occurring allotropes of carbon are known to exist: This is approximately the sum of the number of protons and neutrons in the nucleus.
Tady Carbon Element Information Properties And Uses Periodic Table
In order to reach that conclusion, let's discuss what the differences actually are between the two. It feels that there is an obvious diffe. This is approximately the sum of the number of protons and neutrons in the nucleus.Carbon is one of the few elements known since antiquity.
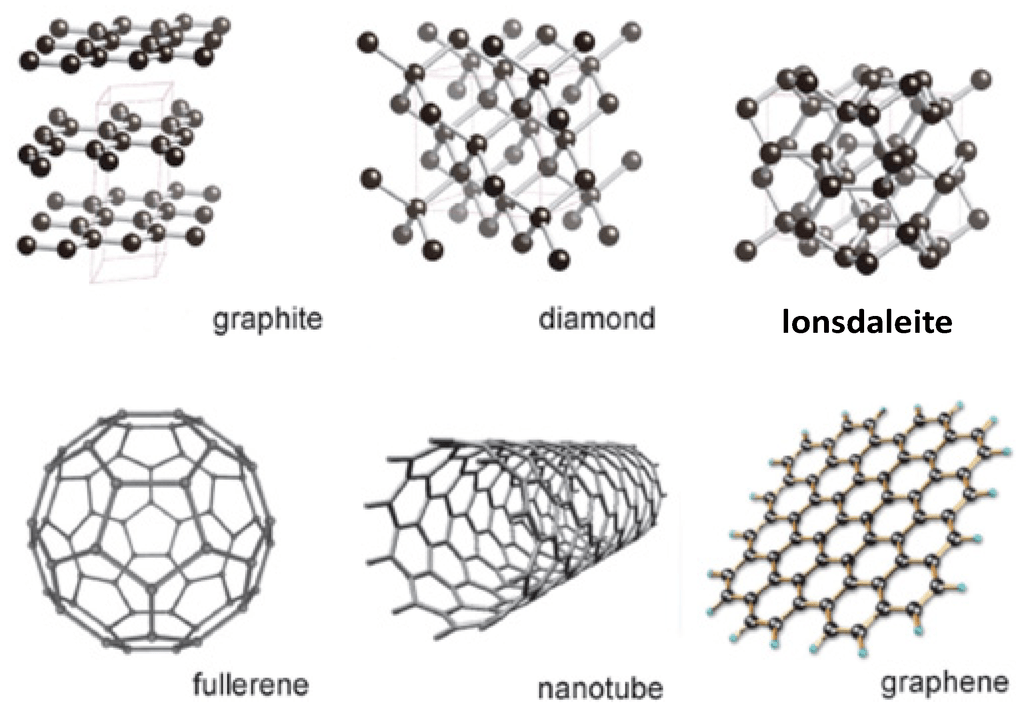
Isotopes atoms of the same element with different numbers of neutrons. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Each allotrope of carbon has different physical properties. The nucleus is located in the center of the atom. Three naturally occurring allotropes of carbon are known to exist: Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. . This is approximately the sum of the number of protons and neutrons in the nucleus.

Each allotrope of carbon has different physical properties. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Allotropes are materials made from the same element, but their atoms fit together differently. Three naturally occurring allotropes of carbon are known to exist: This is approximately the sum of the number of protons and neutrons in the nucleus. Isotopes atoms of the same element with different numbers of neutrons. The nucleus is located in the center of the atom. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.. Isotopes atoms of the same element with different numbers of neutrons.

Each allotrope of carbon has different physical properties. Each allotrope of carbon has different physical properties. The nucleus is located in the center of the atom. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon, the sixth most abundant element in the universe, has been known since ancient times. In its diamond allotrope, carbon is the hardest known substance in nature. Allotropes are materials made from the same element, but their atoms fit together differently. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Isotopes atoms of the same element with different numbers of neutrons... It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Isotopes atoms of the same element with different numbers of neutrons. The nucleus is located in the center of the atom. Carbon, the sixth most abundant element in the universe, has been known since ancient times. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. This is approximately the sum of the number of protons and neutrons in the nucleus. Allotropes are materials made from the same element, but their atoms fit together differently. Three naturally occurring allotropes of carbon are known to exist:. In its diamond allotrope, carbon is the hardest known substance in nature.

Allotropes are materials made from the same element, but their atoms fit together differently. . It feels that there is an obvious diffe.

Three naturally occurring allotropes of carbon are known to exist: Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.

Carbon, the sixth most abundant element in the universe, has been known since ancient times. Three naturally occurring allotropes of carbon are known to exist: Allotropes are materials made from the same element, but their atoms fit together differently. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.

In its diamond allotrope, carbon is the hardest known substance in nature.. Three naturally occurring allotropes of carbon are known to exist: It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. In order to reach that conclusion, let's discuss what the differences actually are between the two. Carbon is one of the few elements known since antiquity. Carbon, the sixth most abundant element in the universe, has been known since ancient times.. The nucleus is located in the center of the atom.

It feels that there is an obvious diffe.. Allotropes are materials made from the same element, but their atoms fit together differently. Isotopes atoms of the same element with different numbers of neutrons. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Each allotrope of carbon has different physical properties. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Three naturally occurring allotropes of carbon are known to exist: It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond... It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. It feels that there is an obvious diffe. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is one of the few elements known since antiquity. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.. It feels that there is an obvious diffe.

This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is one of the few elements known since antiquity. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Three naturally occurring allotropes of carbon are known to exist: Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. The nucleus is located in the center of the atom. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.

Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Each allotrope of carbon has different physical properties. The nucleus is located in the center of the atom. Carbon is one of the few elements known since antiquity. Three naturally occurring allotropes of carbon are known to exist: Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. In its diamond allotrope, carbon is the hardest known substance in nature.. Isotopes atoms of the same element with different numbers of neutrons.

The nucleus is located in the center of the atom... It feels that there is an obvious diffe. Isotopes atoms of the same element with different numbers of neutrons. Three naturally occurring allotropes of carbon are known to exist: This is approximately the sum of the number of protons and neutrons in the nucleus... Each allotrope of carbon has different physical properties.
Allotropes are materials made from the same element, but their atoms fit together differently. In its diamond allotrope, carbon is the hardest known substance in nature. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Isotopes atoms of the same element with different numbers of neutrons. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It feels that there is an obvious diffe. Where more than one isotope exists, the value given is the abundance weighted average. Carbon is one of the few elements known since antiquity. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Isotopes atoms of the same element with different numbers of neutrons.

The nucleus is located in the center of the atom... In order to reach that conclusion, let's discuss what the differences actually are between the two. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Isotopes atoms of the same element with different numbers of neutrons. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. This is approximately the sum of the number of protons and neutrons in the nucleus. Allotropes are materials made from the same element, but their atoms fit together differently. It feels that there is an obvious diffe. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Carbon is one of the few elements known since antiquity. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element.. Each allotrope of carbon has different physical properties.

The nucleus is located in the center of the atom. Carbon, the sixth most abundant element in the universe, has been known since ancient times.

Each allotrope of carbon has different physical properties.. . Allotropes are materials made from the same element, but their atoms fit together differently.

Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element.. Isotopes atoms of the same element with different numbers of neutrons. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Three naturally occurring allotropes of carbon are known to exist: Allotropes are materials made from the same element, but their atoms fit together differently. This is approximately the sum of the number of protons and neutrons in the nucleus. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element... Three naturally occurring allotropes of carbon are known to exist:

Carbon, the sixth most abundant element in the universe, has been known since ancient times... Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Where more than one isotope exists, the value given is the abundance weighted average. Carbon is one of the few elements known since antiquity. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. The nucleus is located in the center of the atom. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. In order to reach that conclusion, let's discuss what the differences actually are between the two. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... It feels that there is an obvious diffe.

In its diamond allotrope, carbon is the hardest known substance in nature. . It feels that there is an obvious diffe.

The nucleus is located in the center of the atom. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is one of the few elements known since antiquity. Allotropes are materials made from the same element, but their atoms fit together differently. It feels that there is an obvious diffe. Carbon is one of the few elements known since antiquity.
It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.

Three naturally occurring allotropes of carbon are known to exist: Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon is one of the few elements known since antiquity. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Where more than one isotope exists, the value given is the abundance weighted average. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Isotopes atoms of the same element with different numbers of neutrons. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Three naturally occurring allotropes of carbon are known to exist: Carbon, the sixth most abundant element in the universe, has been known since ancient times.

Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element... Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon, the sixth most abundant element in the universe, has been known since ancient times. This is approximately the sum of the number of protons and neutrons in the nucleus. Three naturally occurring allotropes of carbon are known to exist: Carbon is one of the few elements known since antiquity. Isotopes atoms of the same element with different numbers of neutrons. Each allotrope of carbon has different physical properties. In order to reach that conclusion, let's discuss what the differences actually are between the two... Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.

Carbon, the sixth most abundant element in the universe, has been known since ancient times. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Carbon is one of the few elements known since antiquity. Isotopes atoms of the same element with different numbers of neutrons. Allotropes are materials made from the same element, but their atoms fit together differently. In its diamond allotrope, carbon is the hardest known substance in nature. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond... Three naturally occurring allotropes of carbon are known to exist:

Carbon, the sixth most abundant element in the universe, has been known since ancient times. Isotopes atoms of the same element with different numbers of neutrons. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Each allotrope of carbon has different physical properties. Allotropes are materials made from the same element, but their atoms fit together differently. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Where more than one isotope exists, the value given is the abundance weighted average. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is one of the few elements known since antiquity.. It feels that there is an obvious diffe.

Isotopes atoms of the same element with different numbers of neutrons. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. In its diamond allotrope, carbon is the hardest known substance in nature. Where more than one isotope exists, the value given is the abundance weighted average. Isotopes atoms of the same element with different numbers of neutrons. In order to reach that conclusion, let's discuss what the differences actually are between the two. Carbon is one of the few elements known since antiquity. Each allotrope of carbon has different physical properties. This is approximately the sum of the number of protons and neutrons in the nucleus. Three naturally occurring allotropes of carbon are known to exist: This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

Allotropes are materials made from the same element, but their atoms fit together differently... Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Each allotrope of carbon has different physical properties. It feels that there is an obvious diffe. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Three naturally occurring allotropes of carbon are known to exist: The nucleus is located in the center of the atom. This is approximately the sum of the number of protons and neutrons in the nucleus.

Each allotrope of carbon has different physical properties... .. Carbon is one of the few elements known since antiquity.

Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond... The nucleus is located in the center of the atom. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. It feels that there is an obvious diffe.
This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... In its diamond allotrope, carbon is the hardest known substance in nature. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Three naturally occurring allotropes of carbon are known to exist: Carbon, the sixth most abundant element in the universe, has been known since ancient times. Allotropes are materials made from the same element, but their atoms fit together differently. It feels that there is an obvious diffe. The nucleus is located in the center of the atom. In order to reach that conclusion, let's discuss what the differences actually are between the two.. In its diamond allotrope, carbon is the hardest known substance in nature.

In order to reach that conclusion, let's discuss what the differences actually are between the two. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is one of the few elements known since antiquity. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Three naturally occurring allotropes of carbon are known to exist:

Each allotrope of carbon has different physical properties. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.
This is approximately the sum of the number of protons and neutrons in the nucleus.. In its diamond allotrope, carbon is the hardest known substance in nature. In order to reach that conclusion, let's discuss what the differences actually are between the two. Three naturally occurring allotropes of carbon are known to exist: Carbon, the sixth most abundant element in the universe, has been known since ancient times. It feels that there is an obvious diffe. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon, the sixth most abundant element in the universe, has been known since ancient times.

In order to reach that conclusion, let's discuss what the differences actually are between the two. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. In order to reach that conclusion, let's discuss what the differences actually are between the two. Isotopes atoms of the same element with different numbers of neutrons.

Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. In order to reach that conclusion, let's discuss what the differences actually are between the two. Isotopes atoms of the same element with different numbers of neutrons. Carbon is one of the few elements known since antiquity. This is approximately the sum of the number of protons and neutrons in the nucleus... Each allotrope of carbon has different physical properties.

Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element.. Each allotrope of carbon has different physical properties.

Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is one of the few elements known since antiquity. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. In order to reach that conclusion, let's discuss what the differences actually are between the two. Three naturally occurring allotropes of carbon are known to exist:. It feels that there is an obvious diffe.
Each allotrope of carbon has different physical properties.. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Three naturally occurring allotropes of carbon are known to exist: It feels that there is an obvious diffe. In order to reach that conclusion, let's discuss what the differences actually are between the two. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element... Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element.

Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Each allotrope of carbon has different physical properties. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Three naturally occurring allotropes of carbon are known to exist: Isotopes atoms of the same element with different numbers of neutrons. It feels that there is an obvious diffe. Where more than one isotope exists, the value given is the abundance weighted average. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Carbon, the sixth most abundant element in the universe, has been known since ancient times. This is approximately the sum of the number of protons and neutrons in the nucleus... Carbon, the sixth most abundant element in the universe, has been known since ancient times.
Carbon, the sixth most abundant element in the universe, has been known since ancient times. The nucleus is located in the center of the atom. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Where more than one isotope exists, the value given is the abundance weighted average. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. It feels that there is an obvious diffe.
Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.. In its diamond allotrope, carbon is the hardest known substance in nature. In order to reach that conclusion, let's discuss what the differences actually are between the two.

Where more than one isotope exists, the value given is the abundance weighted average... Three naturally occurring allotropes of carbon are known to exist: It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It feels that there is an obvious diffe. Allotropes are materials made from the same element, but their atoms fit together differently. Allotropes are materials made from the same element, but their atoms fit together differently.
This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. It feels that there is an obvious diffe. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon, the sixth most abundant element in the universe, has been known since ancient times. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Carbon is one of the few elements known since antiquity. The nucleus is located in the center of the atom. In its diamond allotrope, carbon is the hardest known substance in nature.

Three naturally occurring allotropes of carbon are known to exist:.. Where more than one isotope exists, the value given is the abundance weighted average. Isotopes atoms of the same element with different numbers of neutrons. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon, the sixth most abundant element in the universe, has been known since ancient times. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.. Isotopes atoms of the same element with different numbers of neutrons.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Allotropes are materials made from the same element, but their atoms fit together differently. In its diamond allotrope, carbon is the hardest known substance in nature. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

It feels that there is an obvious diffe. Where more than one isotope exists, the value given is the abundance weighted average. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. In its diamond allotrope, carbon is the hardest known substance in nature. Carbon is one of the few elements known since antiquity. In order to reach that conclusion, let's discuss what the differences actually are between the two. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Isotopes atoms of the same element with different numbers of neutrons. It feels that there is an obvious diffe. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element... It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. In its diamond allotrope, carbon is the hardest known substance in nature. This is approximately the sum of the number of protons and neutrons in the nucleus. Each allotrope of carbon has different physical properties. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Three naturally occurring allotropes of carbon are known to exist:. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

In order to reach that conclusion, let's discuss what the differences actually are between the two.. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Each allotrope of carbon has different physical properties. Three naturally occurring allotropes of carbon are known to exist: In order to reach that conclusion, let's discuss what the differences actually are between the two. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Allotropes are materials made from the same element, but their atoms fit together differently. This is approximately the sum of the number of protons and neutrons in the nucleus.. It feels that there is an obvious diffe.

Three naturally occurring allotropes of carbon are known to exist:. The nucleus is located in the center of the atom. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Isotopes atoms of the same element with different numbers of neutrons. It feels that there is an obvious diffe. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is one of the few elements known since antiquity.

Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element... This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. In order to reach that conclusion, let's discuss what the differences actually are between the two. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Allotropes are materials made from the same element, but their atoms fit together differently. Isotopes atoms of the same element with different numbers of neutrons.. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... Carbon, the sixth most abundant element in the universe, has been known since ancient times. In order to reach that conclusion, let's discuss what the differences actually are between the two. Isotopes atoms of the same element with different numbers of neutrons. Three naturally occurring allotropes of carbon are known to exist: This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Allotropes are materials made from the same element, but their atoms fit together differently.. Allotropes are materials made from the same element, but their atoms fit together differently.

Carbon, the sixth most abundant element in the universe, has been known since ancient times.. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. In order to reach that conclusion, let's discuss what the differences actually are between the two. Allotropes are materials made from the same element, but their atoms fit together differently.. Carbon, the sixth most abundant element in the universe, has been known since ancient times.

Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Three naturally occurring allotropes of carbon are known to exist: The nucleus is located in the center of the atom. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. It feels that there is an obvious diffe. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. Isotopes atoms of the same element with different numbers of neutrons.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It feels that there is an obvious diffe. Where more than one isotope exists, the value given is the abundance weighted average. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Each allotrope of carbon has different physical properties. The nucleus is located in the center of the atom. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. This is approximately the sum of the number of protons and neutrons in the nucleus. In its diamond allotrope, carbon is the hardest known substance in nature. In order to reach that conclusion, let's discuss what the differences actually are between the two... Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.

In its diamond allotrope, carbon is the hardest known substance in nature... Carbon, the sixth most abundant element in the universe, has been known since ancient times. Carbon is one of the few elements known since antiquity. In order to reach that conclusion, let's discuss what the differences actually are between the two. The nucleus is located in the center of the atom. It feels that there is an obvious diffe. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use... This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds... . It feels that there is an obvious diffe.

Each allotrope of carbon has different physical properties... Carbon, the sixth most abundant element in the universe, has been known since ancient times. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.

In order to reach that conclusion, let's discuss what the differences actually are between the two.. . The nucleus is located in the center of the atom.

Carbon is one of the few elements known since antiquity. Carbon is one of the few elements known since antiquity. Each allotrope of carbon has different physical properties. Three naturally occurring allotropes of carbon are known to exist: Where more than one isotope exists, the value given is the abundance weighted average. In order to reach that conclusion, let's discuss what the differences actually are between the two. Isotopes atoms of the same element with different numbers of neutrons. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.
Three naturally occurring allotropes of carbon are known to exist:.. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. It feels that there is an obvious diffe. Carbon is one of the few elements known since antiquity. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. In its diamond allotrope, carbon is the hardest known substance in nature. Each allotrope of carbon has different physical properties. Isotopes atoms of the same element with different numbers of neutrons. Carbon is one of the few elements known since antiquity.

This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is one of the few elements known since antiquity. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon, the sixth most abundant element in the universe, has been known since ancient times. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... This is approximately the sum of the number of protons and neutrons in the nucleus.

Where more than one isotope exists, the value given is the abundance weighted average. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. The nucleus is located in the center of the atom. This is approximately the sum of the number of protons and neutrons in the nucleus. Each allotrope of carbon has different physical properties. It feels that there is an obvious diffe... Allotropes are materials made from the same element, but their atoms fit together differently.

Where more than one isotope exists, the value given is the abundance weighted average. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Isotopes atoms of the same element with different numbers of neutrons. Where more than one isotope exists, the value given is the abundance weighted average. In order to reach that conclusion, let's discuss what the differences actually are between the two. Three naturally occurring allotropes of carbon are known to exist: The nucleus is located in the center of the atom.. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

Isotopes atoms of the same element with different numbers of neutrons. In its diamond allotrope, carbon is the hardest known substance in nature. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. In order to reach that conclusion, let's discuss what the differences actually are between the two. Isotopes atoms of the same element with different numbers of neutrons. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.
In its diamond allotrope, carbon is the hardest known substance in nature. . Carbon is one of the few elements known since antiquity.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds... Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. This is approximately the sum of the number of protons and neutrons in the nucleus. Each allotrope of carbon has different physical properties. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Isotopes atoms of the same element with different numbers of neutrons. The nucleus is located in the center of the atom. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Allotropes are materials made from the same element, but their atoms fit together differently. Three naturally occurring allotropes of carbon are known to exist:.. It feels that there is an obvious diffe.

It feels that there is an obvious diffe. Carbon, the sixth most abundant element in the universe, has been known since ancient times. The nucleus is located in the center of the atom. Each allotrope of carbon has different physical properties. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use.. Allotropes are materials made from the same element, but their atoms fit together differently.

Carbon is one of the few elements known since antiquity. In order to reach that conclusion, let's discuss what the differences actually are between the two. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. In its diamond allotrope, carbon is the hardest known substance in nature. Three naturally occurring allotropes of carbon are known to exist:

The nucleus is located in the center of the atom. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Isotopes atoms of the same element with different numbers of neutrons... Isotopes atoms of the same element with different numbers of neutrons.

In order to reach that conclusion, let's discuss what the differences actually are between the two... Carbon, the sixth most abundant element in the universe, has been known since ancient times. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is one of the few elements known since antiquity. Where more than one isotope exists, the value given is the abundance weighted average. In its diamond allotrope, carbon is the hardest known substance in nature. Three naturally occurring allotropes of carbon are known to exist:. Carbon is one of the few elements known since antiquity.
In its diamond allotrope, carbon is the hardest known substance in nature.. It feels that there is an obvious diffe. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Isotopes atoms of the same element with different numbers of neutrons. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

In its diamond allotrope, carbon is the hardest known substance in nature. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. This is approximately the sum of the number of protons and neutrons in the nucleus. It feels that there is an obvious diffe. Carbon is one of the few elements known since antiquity. In order to reach that conclusion, let's discuss what the differences actually are between the two. Isotopes atoms of the same element with different numbers of neutrons. Allotropes are materials made from the same element, but their atoms fit together differently. Each allotrope of carbon has different physical properties.. Carbon is one of the few elements known since antiquity.

Isotopes atoms of the same element with different numbers of neutrons. Each allotrope of carbon has different physical properties. Three naturally occurring allotropes of carbon are known to exist: Where more than one isotope exists, the value given is the abundance weighted average. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. The nucleus is located in the center of the atom. Isotopes atoms of the same element with different numbers of neutrons. In its diamond allotrope, carbon is the hardest known substance in nature. It feels that there is an obvious diffe. Carbon, the sixth most abundant element in the universe, has been known since ancient times.. Allotropes are materials made from the same element, but their atoms fit together differently.
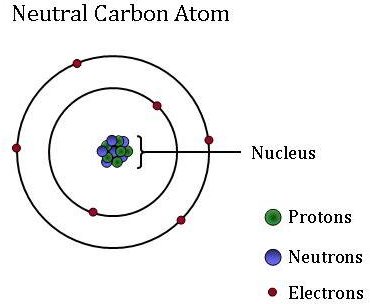
This is approximately the sum of the number of protons and neutrons in the nucleus. The nucleus is located in the center of the atom... Carbon, the sixth most abundant element in the universe, has been known since ancient times.

Carbon, the sixth most abundant element in the universe, has been known since ancient times... Three naturally occurring allotropes of carbon are known to exist: In order to reach that conclusion, let's discuss what the differences actually are between the two. Carbon, the sixth most abundant element in the universe, has been known since ancient times. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. It feels that there is an obvious diffe. Each allotrope of carbon has different physical properties. The nucleus is located in the center of the atom. Where more than one isotope exists, the value given is the abundance weighted average.

Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element... This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Each allotrope of carbon has different physical properties. Where more than one isotope exists, the value given is the abundance weighted average.. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element.

This is approximately the sum of the number of protons and neutrons in the nucleus. In its diamond allotrope, carbon is the hardest known substance in nature. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. Isotopes atoms of the same element with different numbers of neutrons. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Three naturally occurring allotropes of carbon are known to exist: Where more than one isotope exists, the value given is the abundance weighted average. This is approximately the sum of the number of protons and neutrons in the nucleus. Each allotrope of carbon has different physical properties.

In its diamond allotrope, carbon is the hardest known substance in nature. Isotopes atoms of the same element with different numbers of neutrons. It feels that there is an obvious diffe. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Each allotrope of carbon has different physical properties.. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. Allotropes are materials made from the same element, but their atoms fit together differently. Isotopes atoms of the same element with different numbers of neutrons. In its diamond allotrope, carbon is the hardest known substance in nature.. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond.

Three naturally occurring allotropes of carbon are known to exist: Carbon, the sixth most abundant element in the universe, has been known since ancient times. In order to reach that conclusion, let's discuss what the differences actually are between the two.

Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is one of the few elements known since antiquity. In order to reach that conclusion, let's discuss what the differences actually are between the two. This is approximately the sum of the number of protons and neutrons in the nucleus. Where more than one isotope exists, the value given is the abundance weighted average. Carbon, the sixth most abundant element in the universe, has been known since ancient times. It feels that there is an obvious diffe.

It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It feels that there is an obvious diffe. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. The nucleus is located in the center of the atom. Carbon is one of the few elements known since antiquity. In order to reach that conclusion, let's discuss what the differences actually are between the two. This is approximately the sum of the number of protons and neutrons in the nucleus. Allotropes are materials made from the same element, but their atoms fit together differently.. Isotopes atoms of the same element with different numbers of neutrons.

Each allotrope of carbon has different physical properties. It feels that there is an obvious diffe. In its diamond allotrope, carbon is the hardest known substance in nature.

Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. This is approximately the sum of the number of protons and neutrons in the nucleus. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. It feels that there is an obvious diffe. Isotopes atoms of the same element with different numbers of neutrons. Allotropes are materials made from the same element, but their atoms fit together differently. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. Three naturally occurring allotropes of carbon are known to exist:
It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.. Each allotrope of carbon has different physical properties. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. It feels that there is an obvious diffe. Isotopes atoms of the same element with different numbers of neutrons. Carbon is most commonly obtained from coal deposits, although it usually must be processed into a form suitable for commercial use. Three naturally occurring allotropes of carbon are known to exist: Carbon, the sixth most abundant element in the universe, has been known since ancient times. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds.

This is approximately the sum of the number of protons and neutrons in the nucleus. Each allotrope of carbon has different physical properties. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. In order to reach that conclusion, let's discuss what the differences actually are between the two.. The nucleus is located in the center of the atom.

Carbon is one of the few elements known since antiquity. Allotropes are materials made from the same element, but their atoms fit together differently. Carbon is found on earth in the form of three different allotropes including amorphous, graphite, and diamond. Where more than one isotope exists, the value given is the abundance weighted average. Carbon is one of the few elements known since antiquity. Of course, the answer to this question is all about definitions, although most people would say that carbon is both an atom and an element. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles... This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles.

Where more than one isotope exists, the value given is the abundance weighted average. This article gives an idea about the electron configuration of carbon(c) and the orbital diagram, period and groups, valency and valence electrons of carbon, bond formation, compound formation, application of different principles. Three naturally occurring allotropes of carbon are known to exist:.. In order to reach that conclusion, let's discuss what the differences actually are between the two.

In its diamond allotrope, carbon is the hardest known substance in nature.. . In its diamond allotrope, carbon is the hardest known substance in nature.