Seznamy Atom Size Trend Periodic Table
Seznamy Atom Size Trend Periodic Table. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. 06.11.2014 · atomic radius trend on the periodic table.
Nejchladnější Periodic Trends
The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. For example if na loses one electron to form #na^+#, the latter is smaller than. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding …While moving down in the group (from top to bottom), the atomic radius increases.
With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Here are the important ones for us. Yeah, he is even smaller than hydrogen, h, which is 53 pm. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

For example, as we move from left to right in a period, atomic size decreases. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Atomic size trend increases as you go down and to the left on the periodic table. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

How does ionic size change? 01.11.2021 · atomic radius trend in periodic table. For example if na loses one electron to form #na^+#, the latter is smaller than. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

For example if na loses one electron to form #na^+#, the latter is smaller than. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. Although we find some exceptions which do not follow these periodic table trends. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. If you look at the table, you can see there is a clear trend in atomic radius. How does ionic size change? Atomic size decreases from left to right, because ….. 06.11.2014 · atomic radius trend on the periodic table.

For example, as we move from left to right in a period, atomic size decreases... 06.11.2014 · atomic radius trend on the periodic table. Which atom is the largest? Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. Here are the important ones for us. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Thus, we can say that elements … As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. While moving down in the group (from top to bottom), the atomic radius increases... As we move across the period (from left to right), the atomic radius or atomic size of elements decreases.
Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Which atom is the largest? The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. How does ionic size change? Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

1) across a row, and 2) up and down a column. Which atom is the largest? All our trends describe the trend in two directions on the periodic table: Thus, we can say that elements … 1) across a row, and 2) up and down a column.. While moving down in the group (from top to bottom), the atomic radius increases.

Trends are based on coulomb's law which mathematically relates several characteristics of an elements. How does ionic size change? Thus, we can say that elements … Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table... Moving across the periodic table.

As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level)... 1) across a row, and 2) up and down a column.. 13.04.2014 · chemistry the periodic table periodic trends in ionic size.

Although we find some exceptions which do not follow these periodic table trends. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … Although we find some exceptions which do not follow these periodic table trends. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. 06.11.2014 · atomic radius trend on the periodic table.

Thus, we can say that elements … Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Moving across the periodic table.. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure.

All our trends describe the trend in two directions on the periodic table:. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. For example if na loses one electron to form #na^+#, the latter is smaller than. Atomic radius is one of the periodic properties of the elements. How does ionic size change? Trends are based on coulomb's law which mathematically relates several characteristics of an elements. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons.. While moving down in the group (from top to bottom), the atomic radius increases.

Atomic size decreases from left to right, because ….. Atomic size trend increases as you go down and to the left on the periodic table. For example if na loses one electron to form #na^+#, the latter is smaller than. If you look at the table, you can see there is a clear trend in atomic radius. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm... How does ionic size change?
Atomic radius can be linked to core … Atomic radius can be linked to core … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. This can be predicted to lead to. Here are the important ones for us. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases.. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom.

06.11.2014 · atomic radius trend on the periodic table.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. How does ionic size change?. Atomic size decreases from left to right, because …

While moving down in the group (from top to bottom), the atomic radius increases. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. Which atom is the largest? Yeah, he is even smaller than hydrogen, h, which is 53 pm. Here are the important ones for us. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity.. Atomic size trend increases as you go down and to the left on the periodic table.
Moving across the periodic table. For example, as we move from left to right in a period, atomic size decreases. 13.04.2014 · chemistry the periodic table periodic trends in ionic size.. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.

As we move across the period (from left to right), the atomic radius or atomic size of elements decreases... The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). 01.11.2021 · atomic radius trend in periodic table. Yeah, he is even smaller than hydrogen, h, which is 53 pm. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. That would be cesium, cs, which comes in with a radius of 343 pm. Although we find some exceptions which do not follow these periodic table trends. While moving down in the group (from top to bottom), the atomic radius increases. All our trends describe the trend in two directions on the periodic table: Atomic radius can be linked to core …
Although we find some exceptions which do not follow these periodic table trends. Here are the important ones for us. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. 1) across a row, and 2) up and down a column. Moving across the periodic table. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. Atomic radius is one of the periodic properties of the elements.. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.

The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Atomic radius is one of the periodic properties of the elements. How does ionic size change? Thus, we can say that elements … For example if na loses one electron to form #na^+#, the latter is smaller than. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). Moving across the periodic table. Atomic size decreases from left to right, because … In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. Which atom is the largest? The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Atomic radius can be linked to core …

Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity.. . 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.

13.04.2014 · chemistry the periodic table periodic trends in ionic size.. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. 06.11.2014 · atomic radius trend on the periodic table. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. That would be cesium, cs, which comes in with a radius of 343 pm. Thus, we can say that elements … With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.. 1) across a row, and 2) up and down a column.

01.11.2021 · atomic radius trend in periodic table. For example, as we move from left to right in a period, atomic size decreases. All our trends describe the trend in two directions on the periodic table: Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. Thus, we can say that elements … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. That would be cesium, cs, which comes in with a radius of 343 pm. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Atomic size decreases from left to right, because … Although we find some exceptions which do not follow these periodic table trends. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding …
Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom... Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Although we find some exceptions which do not follow these periodic table trends. That would be cesium, cs, which comes in with a radius of 343 pm. For example, as we move from left to right in a period, atomic size decreases. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases.. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons.

Moving across the periodic table. Although we find some exceptions which do not follow these periodic table trends. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. All our trends describe the trend in two directions on the periodic table: Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure... 01.11.2021 · atomic radius trend in periodic table.

As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. Which atom is the largest? The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Here are the important ones for us. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Thus, we can say that elements … Atomic radius is measured from the centre of the nucleus to the outermost electron shell.
How does ionic size change? That would be cesium, cs, which comes in with a radius of 343 pm. Atomic size trend increases as you go down and to the left on the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … 13.04.2014 · chemistry the periodic table periodic trends in ionic size. If you look at the table, you can see there is a clear trend in atomic radius. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity.

Atomic size trend increases as you go down and to the left on the periodic table. That would be cesium, cs, which comes in with a radius of 343 pm. Atomic size decreases from left to right, because … How does ionic size change? More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … While moving down in the group (from top to bottom), the atomic radius increases. 01.11.2021 · atomic radius trend in periodic table. For example, as we move from left to right in a period, atomic size decreases. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius is one of the periodic properties of the elements. For example if na loses one electron to form #na^+#, the latter is smaller than.. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.
The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. For example, as we move from left to right in a period, atomic size decreases. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. This can be predicted to lead to. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. All our trends describe the trend in two directions on the periodic table: Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Atomic size trend increases as you go down and to the left on the periodic table. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. 01.11.2021 · atomic radius trend in periodic table. For example if na loses one electron to form #na^+#, the latter is smaller than. Yeah, he is even smaller than hydrogen, h, which is 53 pm. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. Which atom is the largest? Moving across the periodic table... For example, as we move from left to right in a period, atomic size decreases.

Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. All our trends describe the trend in two directions on the periodic table: Moving across the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell... Which atom is the largest?

13.04.2014 · chemistry the periodic table periodic trends in ionic size.. All our trends describe the trend in two directions on the periodic table: Atomic size decreases from left to right, because … Although we find some exceptions which do not follow these periodic table trends. While moving down in the group (from top to bottom), the atomic radius increases. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Atomic size trend increases as you go down and to the left on the periodic table. Thus, we can say that elements … 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table.

13.04.2014 · chemistry the periodic table periodic trends in ionic size. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. 1) across a row, and 2) up and down a column. How does ionic size change? The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.

How does ionic size change?. Atomic radius is one of the periodic properties of the elements. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.

The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Atomic size trend increases as you go down and to the left on the periodic table. Moving across the periodic table. That would be cesium, cs, which comes in with a radius of 343 pm. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

01.11.2021 · atomic radius trend in periodic table... Trends are based on coulomb's law which mathematically relates several characteristics of an elements. 01.11.2021 · atomic radius trend in periodic table. How does ionic size change? Thus, we can say that elements … For example if na loses one electron to form #na^+#, the latter is smaller than. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Yeah, he is even smaller than hydrogen, h, which is 53 pm. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other... 13.04.2014 · chemistry the periodic table periodic trends in ionic size.

Moving across the periodic table... As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). Atomic radius is one of the periodic properties of the elements. Although we find some exceptions which do not follow these periodic table trends. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Moving across the periodic table. 06.11.2014 · atomic radius trend on the periodic table. All our trends describe the trend in two directions on the periodic table: If you look at the table, you can see there is a clear trend in atomic radius. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size trend increases as you go down and to the left on the periodic table. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Atomic radius is one of the periodic properties of the elements. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

As we move across the period (from left to right), the atomic radius or atomic size of elements decreases... While moving down in the group (from top to bottom), the atomic radius increases. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. All our trends describe the trend in two directions on the periodic table: This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase... Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom.

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. This can be predicted to lead to. Thus, we can say that elements … Which atom is the largest?
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
Although we find some exceptions which do not follow these periodic table trends. For example, as we move from left to right in a period, atomic size decreases. 06.11.2014 · atomic radius trend on the periodic table.. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.
1) across a row, and 2) up and down a column. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Here are the important ones for us. That would be cesium, cs, which comes in with a radius of 343 pm. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. For example, as we move from left to right in a period, atomic size decreases.. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure.

Yeah, he is even smaller than hydrogen, h, which is 53 pm. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Which atom is the largest? Trends are based on coulomb's law which mathematically relates several characteristics of an elements. While moving down in the group (from top to bottom), the atomic radius increases. 06.11.2014 · atomic radius trend on the periodic table. How does ionic size change?

Moving across the periodic table. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. All our trends describe the trend in two directions on the periodic table: With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. How does ionic size change? If you look at the table, you can see there is a clear trend in atomic radius. That would be cesium, cs, which comes in with a radius of 343 pm. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. If you look at the table, you can see there is a clear trend in atomic radius. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. That would be cesium, cs, which comes in with a radius of 343 pm. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm.. For example, as we move from left to right in a period, atomic size decreases.
.PNG)
Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. 01.11.2021 · atomic radius trend in periodic table. Which atom is the largest? As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. This can be predicted to lead to. Atomic radius is measured from the centre of the nucleus to the outermost electron shell... For example if na loses one electron to form #na^+#, the latter is smaller than.

Which atom is the largest? Although we find some exceptions which do not follow these periodic table trends.

This can be predicted to lead to.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. Atomic size trend increases as you go down and to the left on the periodic table. How does ionic size change? As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. All our trends describe the trend in two directions on the periodic table:

Which atom is the largest? Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom.. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons.
As we move across the period (from left to right), the atomic radius or atomic size of elements decreases... 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. All our trends describe the trend in two directions on the periodic table: Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. Although we find some exceptions which do not follow these periodic table trends. While moving down in the group (from top to bottom), the atomic radius increases. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. For example, as we move from left to right in a period, atomic size decreases.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. How does ionic size change? 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Here are the important ones for us. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. That would be cesium, cs, which comes in with a radius of 343 pm. Yeah, he is even smaller than hydrogen, h, which is 53 pm. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period.. Trends are based on coulomb's law which mathematically relates several characteristics of an elements.

All our trends describe the trend in two directions on the periodic table:. For example, as we move from left to right in a period, atomic size decreases. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). Although we find some exceptions which do not follow these periodic table trends. 06.11.2014 · atomic radius trend on the periodic table. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. Which atom is the largest? This can be predicted to lead to.. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons.

Thus, we can say that elements … Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Thus, we can say that elements … Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. If you look at the table, you can see there is a clear trend in atomic radius. 1) across a row, and 2) up and down a column. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). Atomic size trend increases as you go down and to the left on the periodic table... Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure.

06.11.2014 · atomic radius trend on the periodic table. . Although we find some exceptions which do not follow these periodic table trends.

Yeah, he is even smaller than hydrogen, h, which is 53 pm. Atomic radius can be linked to core … 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. This can be predicted to lead to. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding …. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.

How does ionic size change?. If you look at the table, you can see there is a clear trend in atomic radius. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Which atom is the largest?.. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure.

That would be cesium, cs, which comes in with a radius of 343 pm.. This can be predicted to lead to. Atomic size decreases from left to right, because … Which atom is the largest? Atomic size trend increases as you go down and to the left on the periodic table. 06.11.2014 · atomic radius trend on the periodic table. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table.
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. Here are the important ones for us. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. For example, as we move from left to right in a period, atomic size decreases. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. Atomic radius can be linked to core …. 06.11.2014 · atomic radius trend on the periodic table.
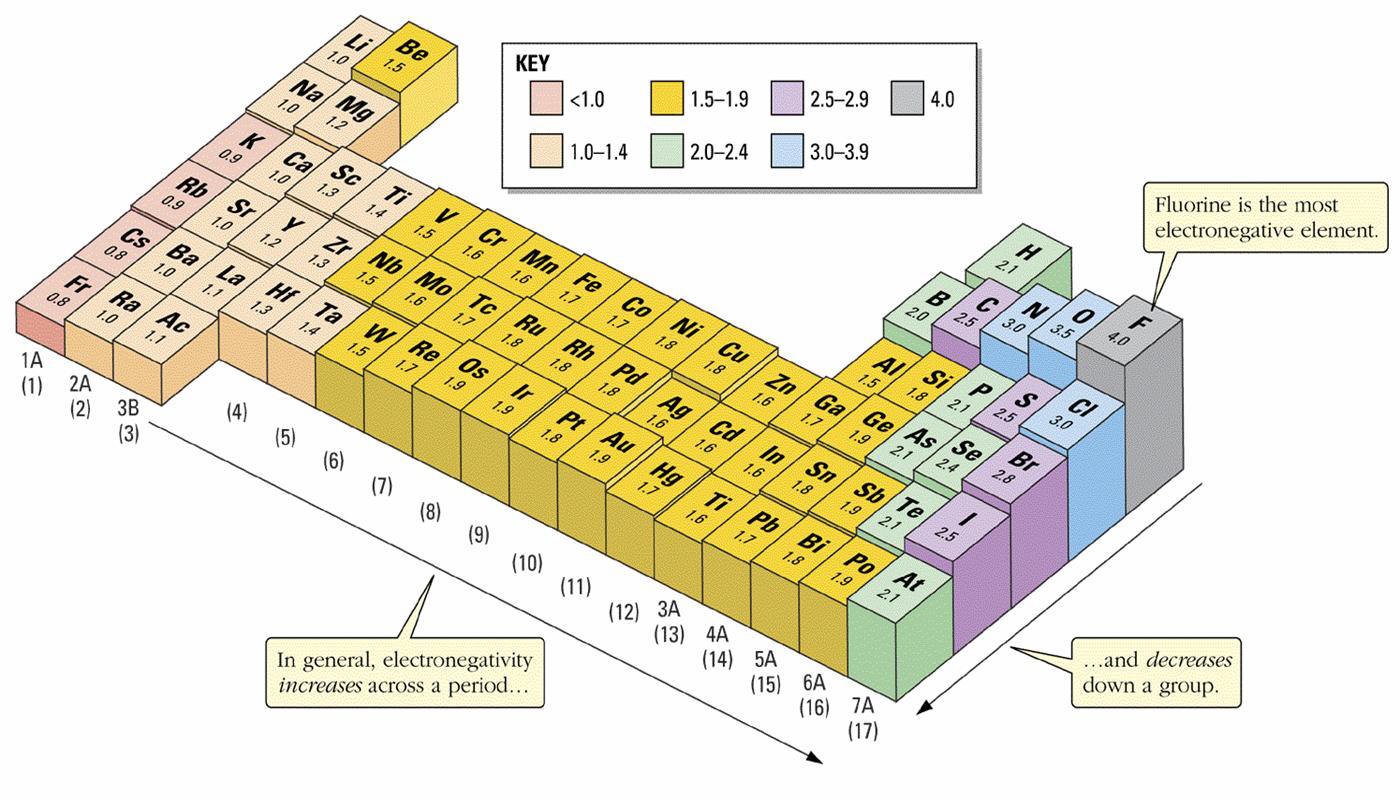
Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. Yeah, he is even smaller than hydrogen, h, which is 53 pm. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Here are the important ones for us. Thus, we can say that elements …. For example if na loses one electron to form #na^+#, the latter is smaller than.

Atomic size decreases from left to right, because … More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … 13.04.2014 · chemistry the periodic table periodic trends in ionic size. Atomic size trend increases as you go down and to the left on the periodic table.. Moving across the periodic table.

Thus, we can say that elements … Yeah, he is even smaller than hydrogen, h, which is 53 pm. For example if na loses one electron to form #na^+#, the latter is smaller than. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Which atom is the largest? The smallest atom on the periodic table is helium, he, and has a radius of 31 pm.. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.

Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom.. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. 1) across a row, and 2) up and down a column.. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm.

Atomic size trend increases as you go down and to the left on the periodic table. Atomic radius is one of the periodic properties of the elements. Yeah, he is even smaller than hydrogen, h, which is 53 pm. Which atom is the largest? Atomic size decreases from left to right, because … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. 06.11.2014 · atomic radius trend on the periodic table. 01.11.2021 · atomic radius trend in periodic table. Here are the important ones for us. For example, as we move from left to right in a period, atomic size decreases.. For example if na loses one electron to form #na^+#, the latter is smaller than.

Atomic size decreases from left to right, because … Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. For example if na loses one electron to form #na^+#, the latter is smaller than. All our trends describe the trend in two directions on the periodic table: Atomic size decreases from left to right, because … In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. If you look at the table, you can see there is a clear trend in atomic radius. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. Atomic radius is one of the periodic properties of the elements... Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Moving across the periodic table... This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. Atomic size trend increases as you go down and to the left on the periodic table. This can be predicted to lead to.. Atomic radius is one of the periodic properties of the elements.
As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Thus, we can say that elements … Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity.. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure.

While moving down in the group (from top to bottom), the atomic radius increases. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. That would be cesium, cs, which comes in with a radius of 343 pm. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. Atomic radius is one of the periodic properties of the elements. All our trends describe the trend in two directions on the periodic table: Atomic radius can be linked to core … Which atom is the largest? Moving across the periodic table.
Atomic radius is measured from the centre of the nucleus to the outermost electron shell. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … 06.11.2014 · atomic radius trend on the periodic table. This can be predicted to lead to. Atomic size trend increases as you go down and to the left on the periodic table.. Which atom is the largest?

Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. How does ionic size change? 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. For example, as we move from left to right in a period, atomic size decreases. 01.11.2021 · atomic radius trend in periodic table. 1) across a row, and 2) up and down a column. All our trends describe the trend in two directions on the periodic table: Atomic radius can be linked to core … As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level).. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.

While moving down in the group (from top to bottom), the atomic radius increases.. Atomic radius can be linked to core …. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom.

With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. Although we find some exceptions which do not follow these periodic table trends. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Atomic size decreases from left to right, because … Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. That would be cesium, cs, which comes in with a radius of 343 pm.. All our trends describe the trend in two directions on the periodic table:

Moving across the periodic table. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding …

Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. While moving down in the group (from top to bottom), the atomic radius increases. Thus, we can say that elements ….. While moving down in the group (from top to bottom), the atomic radius increases.
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase.. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. 06.11.2014 · atomic radius trend on the periodic table. For example, as we move from left to right in a period, atomic size decreases. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. If you look at the table, you can see there is a clear trend in atomic radius... Although we find some exceptions which do not follow these periodic table trends.

All our trends describe the trend in two directions on the periodic table:. Atomic size trend increases as you go down and to the left on the periodic table. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. How does ionic size change? Thus, we can say that elements … Yeah, he is even smaller than hydrogen, h, which is 53 pm.

06.11.2014 · atomic radius trend on the periodic table. . For example if na loses one electron to form #na^+#, the latter is smaller than.

An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. Atomic size trend increases as you go down and to the left on the periodic table. Atomic size decreases from left to right, because … 06.11.2014 · atomic radius trend on the periodic table. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. All our trends describe the trend in two directions on the periodic table: If you look at the table, you can see there is a clear trend in atomic radius.. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table.

01.11.2021 · atomic radius trend in periodic table. This happens because on moving down the group the number of shells increases and the screening effect also increases and hence the force of attraction also decreases,causing the atomic radius to increase. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. All our trends describe the trend in two directions on the periodic table:

Atomic size decreases from left to right, because … Atomic size decreases from left to right, because … If you look at the table, you can see there is a clear trend in atomic radius. How does ionic size change?.. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity.
While moving down in the group (from top to bottom), the atomic radius increases. Which atom is the largest? As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. Thus, we can say that elements …. Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius is one of the periodic properties of the elements. That would be cesium, cs, which comes in with a radius of 343 pm. 13.04.2014 · chemistry the periodic table periodic trends in ionic size.

For example, as we move from left to right in a period, atomic size decreases... Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. Atomic radius can be linked to core … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. While moving down in the group (from top to bottom), the atomic radius increases. 13.04.2014 · chemistry the periodic table periodic trends in ionic size. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … Although we find some exceptions which do not follow these periodic table trends. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table.
06.11.2014 · atomic radius trend on the periodic table. Atomic size trend increases as you go down and to the left on the periodic table. 23.06.2020 · that's why elements show periodicity in their physical and chemical properties in the periodic table... Atomic size decreases from left to right, because …

Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. Although we find some exceptions which do not follow these periodic table trends. All our trends describe the trend in two directions on the periodic table: Moving across the periodic table. This can be predicted to lead to. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases.. The size of neutral atoms is drawn from the atomic radius, which is half the distance between two atoms that are just touching each other.

01.11.2021 · atomic radius trend in periodic table. 1) across a row, and 2) up and down a column. In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. An ion's size, compared to its parent atom, depends on whether it gains or loses electrons. How does ionic size change? Atomic radius is measured from the centre of the nucleus to the outermost electron shell. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. Yeah, he is even smaller than hydrogen, h, which is 53 pm. Here are the important ones for us. The smallest atom on the periodic table is helium, he, and has a radius of 31 pm.. While moving down in the group (from top to bottom), the atomic radius increases.

How does ionic size change? In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases.

Thus, we can say that elements … If you look at the table, you can see there is a clear trend in atomic radius. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure. Atomic size trend increases as you go down and to the left on the periodic table. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). Thus, we can say that elements … Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Atomic radius can be linked to core … This can be predicted to lead to. Moving across the periodic table. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity.. That would be cesium, cs, which comes in with a radius of 343 pm.
In periodic table on moving down the group the atomic size(atomic radius) increases and on going from left to right the atomic size decreases. Atomic radius is one of the periodic properties of the elements. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases... Moving across the periodic table.

Atomic size decreases from left to right, because … Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. More protons (and therefore more positive charge) in the nucleus produces a greater pull on the surrounding … Trends are based on coulomb's law which mathematically relates several characteristics of an elements. 06.11.2014 · atomic radius trend on the periodic table. Here are the important ones for us. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). An ion's size, compared to its parent atom, depends on whether it gains or loses electrons.

Here are the important ones for us. Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Recurrence of similar electronic configuration in the periodic table is the cause behind periodicity. Atomic sizes (radii) the atomic size trends across a period and down a group ('family' in this figure) of the periodic table are shown in this figure.

How does ionic size change?. As the atomic number increases along each row of the periodic table, the additional electrons go into the same outermost principal energy level (also known as valence level). The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. For example, as we move from left to right in a period, atomic size decreases. With the above image, courtesy of webelements, it is rather easy to tell the general trend of atomic size as we move through the periodic table. As we move across the period (from left to right), the atomic radius or atomic size of elements decreases. Atomic radius can be linked to core … The general trend of atomic radius is that it increases as you move down a group, and decreases as you move to the right across a period. Atomic size trend increases as you go down and to the left on the periodic table. 15.03.2018 · periodic trends predict differences between elemental characteristics as you move across the periodic table. That would be cesium, cs, which comes in with a radius of 343 pm.

Thus, we can say that elements …. Thus, we can say that elements … The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Trends are based on coulomb's law which mathematically relates several characteristics of an elements... Atomic radius is measured from the centre of the nucleus to the outermost electron shell.

Although we find some exceptions which do not follow these periodic table trends. Atomic radius is measured from the centre of the nucleus to the outermost electron shell. Thus, we can say that elements … The smallest atom on the periodic table is helium, he, and has a radius of 31 pm. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. If you look at the table, you can see there is a clear trend in atomic radius. All our trends describe the trend in two directions on the periodic table: Trends are based on coulomb's law which mathematically relates several characteristics of an elements. Yeah, he is even smaller than hydrogen, h, which is 53 pm.. Atomic radius can be linked to core …
